CDx Development
Studies have proven that drug development success rates drastically increase with the co-development of an appropriate biomarker beginning at the early stages.
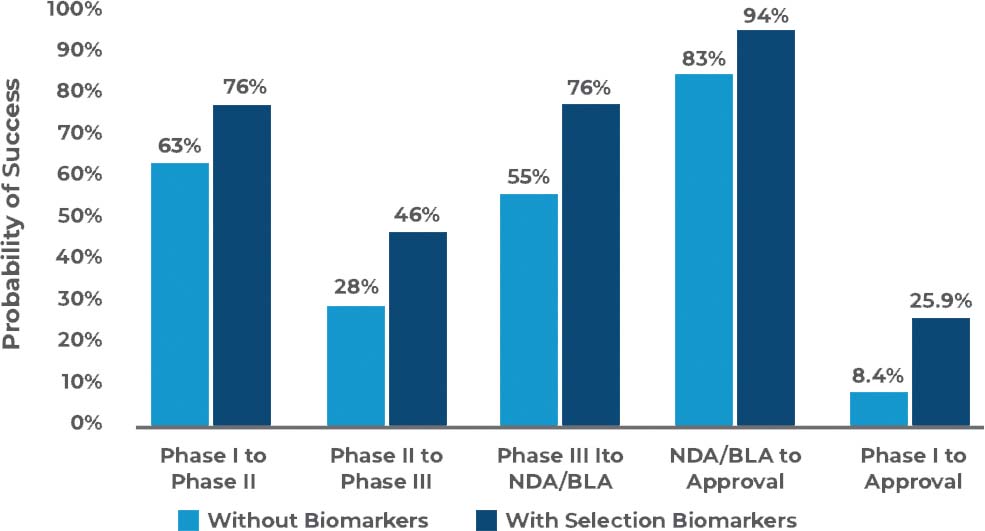
Through our recent partnership with Flagship Biosciences, you can now make confident, more informed decisions during your entire drug development process.
Clinical & CDx Services

Image Analysis
Remove the inherent variability of subjective manual scoring and generate unique cellular patient profiles with data-rich tissue interpretations that incorporate image analysis, machine learning, statistical analysis, and pathologist oversight to help our partners tailor their therapies to targeted patient populations.

Data Reporting
Better understand how your cellular data impacts your drug development with actionable and decision-enabling intelligence. Using machine learning algorithms that analyze whole tissue, tumor/TME, and phenotype-specific data you can now create a powerful profile of your tissues and cohorts to find endpoints resulting in your desired outcomes.
Multiplex Assays
Analyze multiple markers simultaneously across tissue types and eliminate the blind spots caused by single markers that can lead to an incomplete or inaccurate picture of treatment safety and efficacy. Produce more effective therapies with custom and off-the-shelf multiplex panels backed by pathology review and industry leading artificial intelligence.

Regulatory Support
Ensure concise, stepwise regulatory development in a timely and cost-efficient manner. Create a strategic regulatory roadmap for the development of your CDx biomarker with the support of a team who has a deep understanding of the regulatory standards and pathways that drive biomarker development at all stages of the approval process.